VARROMED® – OTC medicine against varroa
555 ml NET VOLUME
Description:
OTC veterinary medicinal product
New in the EU for the control of varroa.
Composition:
1 ml of product contains: Formic acid 5 mg, Oxalic acid dihydrate 44 mg (equivalent to 31.42 mg of anhydrous oxalic acid);
Excipients: Sulphite ammonia caramel (E150d), Sucrose syrup, Propolis extract 20%, Star anise oil, Lemon oil, Citric acid monohydrate, Purified water.
Pharmaceutical form:
Ready-to-use suspension for pouring into the hive, light brown to dark brown in color.
Indications:
Control of varroa (Varroa destructor) in honeybee colonies with and without brood.
Contraindications:
Do not use during the honey flow period.
Dosage and route of application:
In-hive application. Apply the product to the bees in the inter-frame spaces of the nest box. Ensure that the treated bees have sufficient access to drinking water.
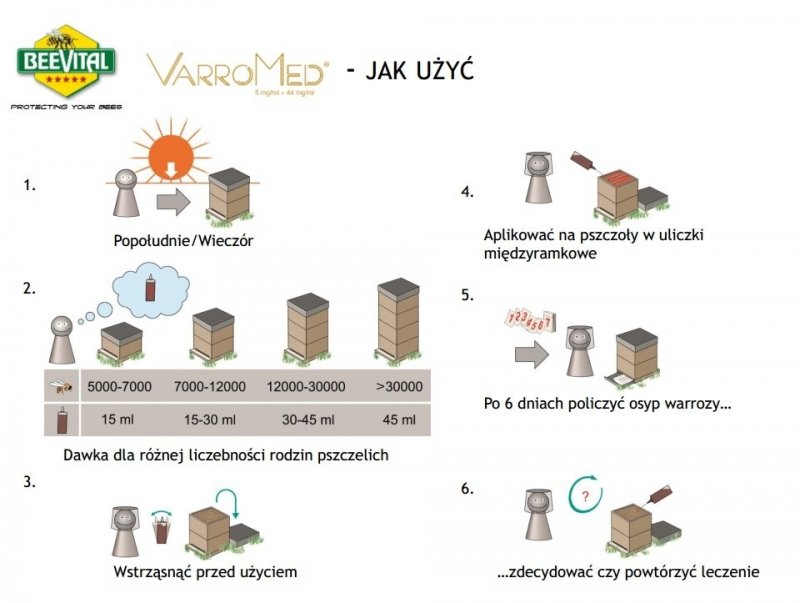
Dosage:
Shake before use. The dose should be carefully adjusted to the size of the bee colony (see dosage table). Determine the size of the bee colony and the number of spaces occupied by the bees to be treated, and then select the required amount of product. The following dosing schedule should be used according to the table:
| Number of bees | 5000 – 7000 | 7000 – 12000 | 12000 – 30000 | >30000 |
| VarroMed (ml) | 15 | 15 to 30 | 30 to 45 | 45 |
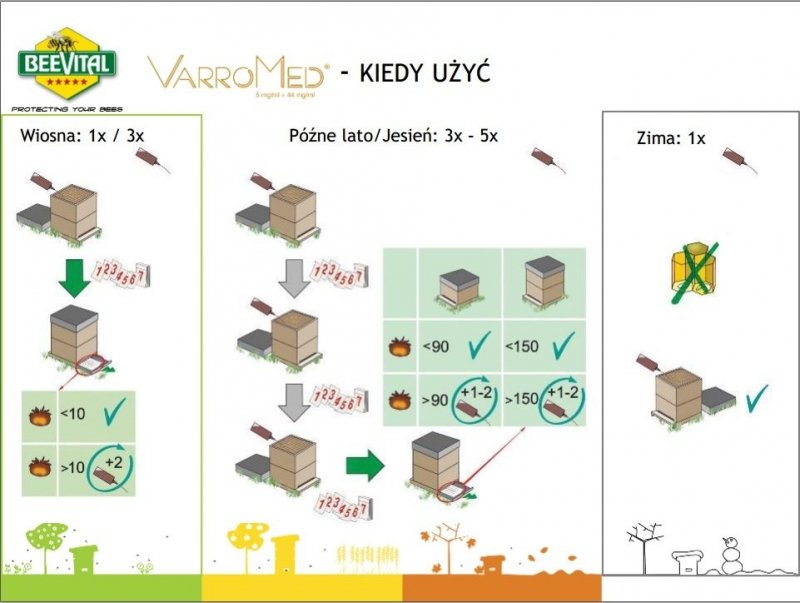
Recommendations for proper application:
The product should be used primarily during periods of low bee flight activity (late afternoon and evening). To avoid overdosing individual bees, try to distribute the product evenly across the bees, especially in the winter cluster. VarroMed should not be used during the nectar flow period or when there are honeycombs in the hive. Before use, warm the product to a temperature of 25°C to 35°C and shake well.
Before application, it is recommended to remove wax bridges between the top slats of the frames. Frames should not be lifted during application and for approximately one week after the last treatment cycle. To determine the level of mite infestation in the hive, monitor mite mortality: monitor the mite litter on the bottom liner before the first treatment cycle and then for a maximum of 6 days after each treatment cycle. Treat all bee colonies in the same location simultaneously to minimize the risk of mite reinfestation.
Overdose (symptoms, treatment):
After application of a 10% solution of oxalic acid dihydrate in a 50% sugar solution, permanent changes in the digestive tract and excretory organs were observed after 72 hours.
Application of a 20% concentration of oxalic acid in a 50% sugar solution resulted in the death of over 60% of the bees. In the event of an accidental overdose (e.g., after a large amount of VarroMed was spilled in the hive), the best remedy is to replace the hive body and clean all visible traces of the suspension from the frames with water.
Interactions with other medicinal products:
Unknown. Avoid concurrent use of other acaricidal products.
Withdrawal period:
Honey: 0 (zero) days.
Special warnings:
VarroMed should be used as part of an integrated varroa control program. Efficacy was tested in hives with low to moderate mite infestation rates.
Shelf life:
The product as packaged for sale: 2 years.
After first opening of the packaging: 30 days.
Special storage precautions:
Do not store above 25°C. Keep the bottle tightly closed. Keep the bottle in the outer cardboard box to protect from light.
Type of immediate packaging:
Cardboard box containing a HDPE (LDPE) bottle with a dropper and a cap (with a tamper-evident seal). The bottle is equipped with a graduation. The box contains one bottle containing 555 ml of suspension.
Name and address of the marketing authorisation holder:
BeeVital GmbH, Wiesenbergstrasse 19, 5164 Seeham, AUSTRIA.
EU marketing authorisation number:
Registered in the Community Register of Medicinal Products under number: EU/2/16/203/001-002. ATC vet code: QP53AG30.
Detailed information is available on the European Medicines Agency website http://www.ema.europa.eu/.
Information about this medicine is provided on the product packaging and on the package leaflet.
Veterinary medicinal product available without a prescription – OTC classification.
 |
We are under the supervision of the Provincial Veterinary Inspectorate in Lublin (Poland) ul. Droga Męczenników Majdanka 50 20-325 Lublin |